Among the 20 standard amino acids that make up proteins, glycine is special. Most amino acids have a chiral center and require consideration of stereoisomers such as D and L forms. However, glycine alone has no optical isomers. The reason is that glycine's side chain is a hydrogen atom.
Substituents at the α-Carbon: Glycine Versus Other Amino Acids
The general structure of an α-amino acid can be expressed as:
H2N-CH(R)-COOH
In most amino acids, the α-carbon is bonded to four different substituents: an amino group, a carboxyl group, a hydrogen atom, and an R group. Such a carbon becomes a chiral center and possesses enantiomers.
However, in glycine, the R group is hydrogen. Therefore, the substituents bonded to the α-carbon are: amino group, carboxyl group, hydrogen, hydrogen. Because there are two hydrogen atoms, the four substituents are not all different.
PubChem describes glycine as the simplest protein-forming amino acid, with a hydrogen atom as its side chain, and as the only achiral protein-forming amino acid. NCBI Bookshelf also explains that all 20 protein-forming amino acids are L-isomers, with chiral centers at the α-carbon except for glycine.
Comparing Glycine and Alanine
| Amino Acid | Substituents at α-carbon | Chiral Center | Optical Isomers |
|---|---|---|---|
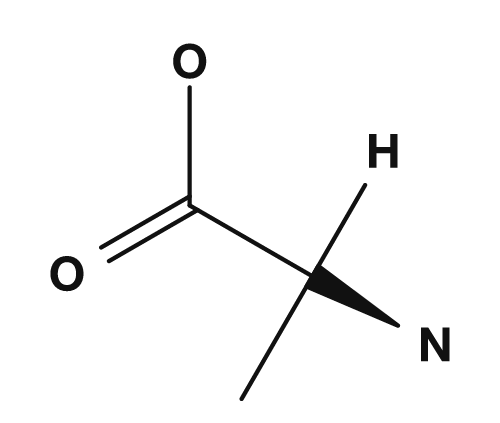
| Alanine (example) | NH2, COOH, H, CH3 |
Yes | D and L forms exist |
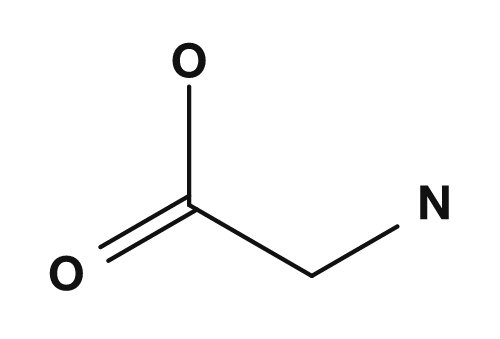
| Glycine | NH2, COOH, H, H |
No | Do not exist |
| Alanine | Glycine |
|---|---|
|
|
Chiral Center and Optical Isomers
Having no chiral center means that glycine has no distinction between D and L forms. In amino acids like alanine and valine, two configurations that are mirror images of each other can be distinguished, but in glycine, because there are two identical substituents around the α-carbon, the mirror image can be superimposed on the original structure. This is why glycine has no optical isomers.
Confirmation from Structural Formulas
This difference becomes clearer when confirmed with structural formulas.
- Alanine: α-carbon bonded to
NH2,COOH,H,CH3→ all four are different → chiral - Glycine: α-carbon bonded to
NH2,COOH,H,H→ two hydrogens → no chiral center
In other words, glycine's special nature can be understood immediately from the structural formula.
Effect on Protein Structure
The small size of glycine also affects protein structure. Because the side chain is only hydrogen, glycine has less steric bulk than other amino acids, and tends to appear at positions in a protein where the backbone can move relatively freely. NCBI Bookshelf's Molecular Biology of the Cell explains that the three-dimensional structure of a protein is determined by its amino acid sequence, and that interactions between side chains stabilize the folding. Glycine's small side chain plays a unique role in such three-dimensional structures.
Glycine is also the simplest amino acid, making it suitable as the first example when learning about the basic structure of amino acids. However, in explanations of chirality, it must be treated as an exception. It is important to understand that most amino acids are chiral, but glycine is different.
Conditions for a Chiral Center
Understanding glycine also helps you naturally understand the conditions for a chiral center.
- The carbon has four bonds (necessary but not sufficient)
- The four substituents are all different (this is the necessary condition)
Glycine does not satisfy condition 2, so it does not become a chiral center. It is not enough simply for a carbon to have four bonds. All four substituents must be different.
Summary
The reason glycine alone has no optical isomers is that its side chain is a hydrogen atom, and the α-carbon has two identical hydrogen atoms bonded to it. Therefore, the α-carbon does not have four different substituents and does not become a chiral center. Glycine is the simplest of the protein-forming amino acids and the only achiral amino acid.