There are 20 standard amino acids that make up proteins. They all share the same basic skeleton, but their side chains (R groups) differ. NCBI Bookshelf lists the 20 proteinogenic amino acids as alanine, arginine, asparagine, aspartic acid, cysteine, glutamic acid, glutamine, glycine, histidine, isoleucine, leucine, lysine, methionine, phenylalanine, proline, serine, threonine, tryptophan, tyrosine, and valine.
When learning the 20 amino acids, it is easier to understand them by classifying them according to the properties of their side chains rather than memorizing the names by rote.
First, here is a list of all 20.
| Category | Main amino acids | Side chain features |
|---|---|---|
| Nonpolar | Glycine, alanine, valine, leucine, isoleucine, proline, methionine | Tend not to mix with water |
| Polar (uncharged) | Serine, threonine, asparagine, glutamine | Tend to participate in hydrogen bonding |
| Acidic | Aspartic acid, glutamic acid | Negatively charged at physiological pH |
| Basic | Lysine, arginine, histidine | Have basic side chains and tend to carry positive charge |
| Aromatic | Phenylalanine, tyrosine, tryptophan | Have aromatic rings |
| Sulfur-containing | Cysteine, methionine | Have a thiol group or thioether |
| Special structure | Proline | Secondary amine, cyclic structure |
| Glycine | Alanine | Valine | Leucine | Isoleucine |
|---|---|---|---|---|
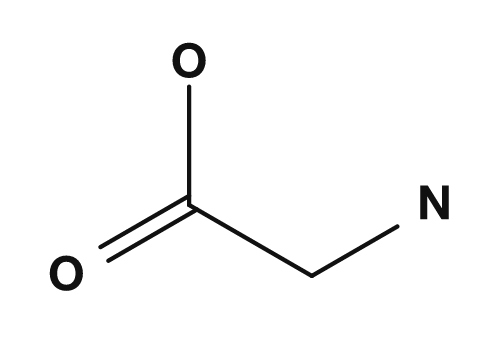
|
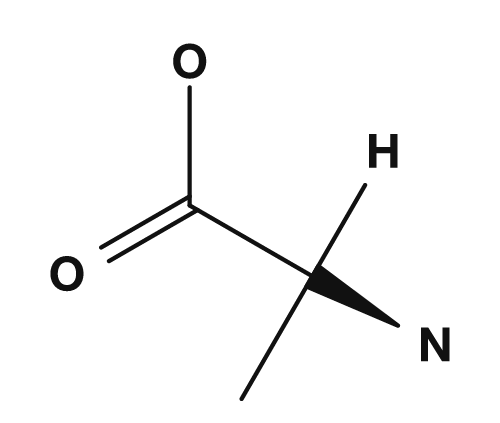
|
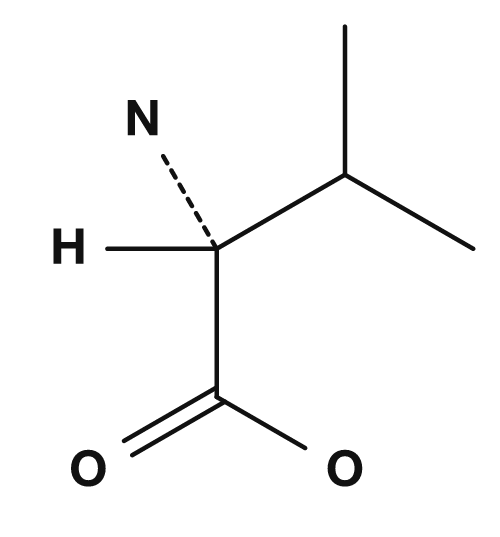
|
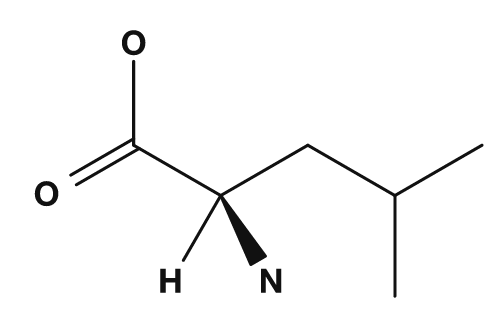
|
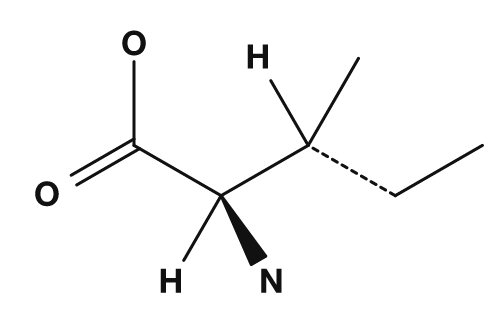
|
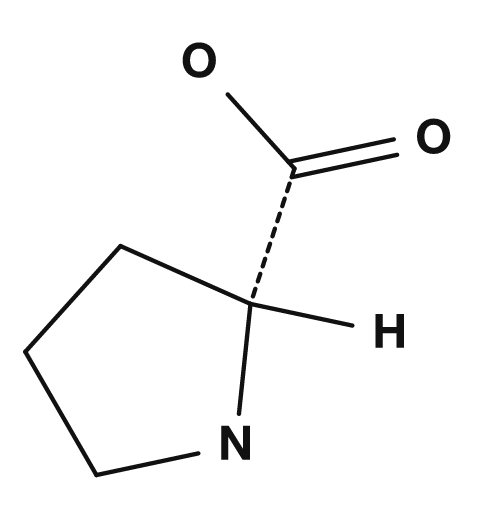
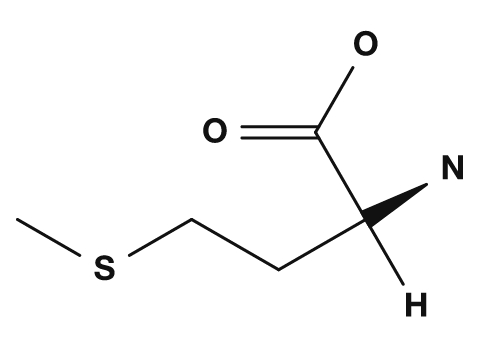
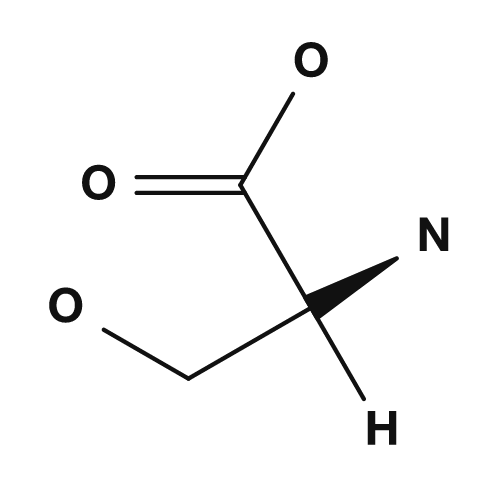
| Proline | Methionine | Serine | Threonine | Cysteine |
|---|---|---|---|---|
|
|
|
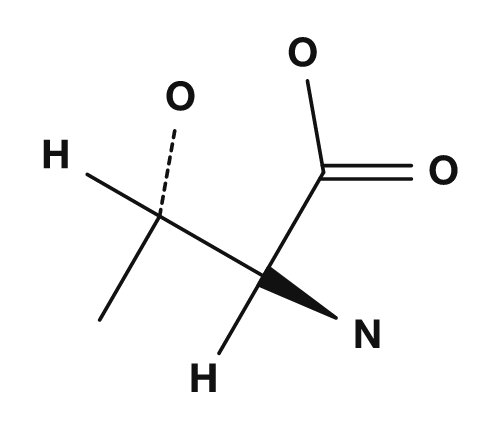
|
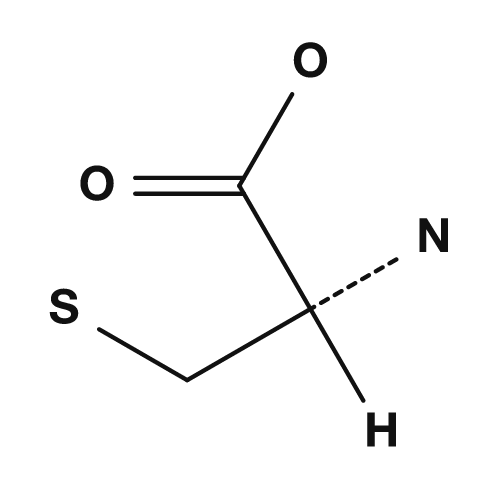
|
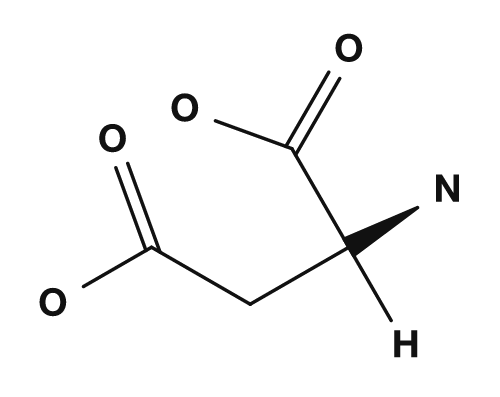
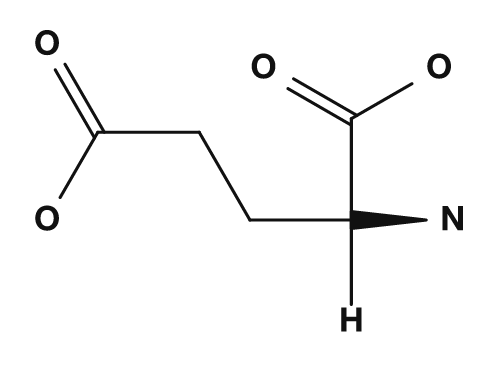
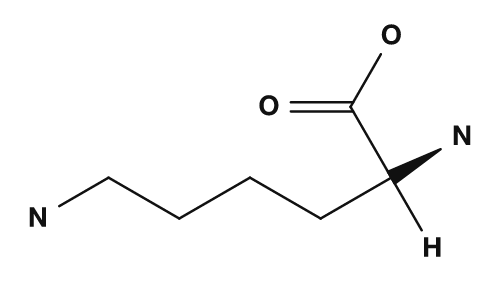
| Asparagine | Glutamine | Aspartic acid | Glutamic acid | Lysine |
|---|---|---|---|---|
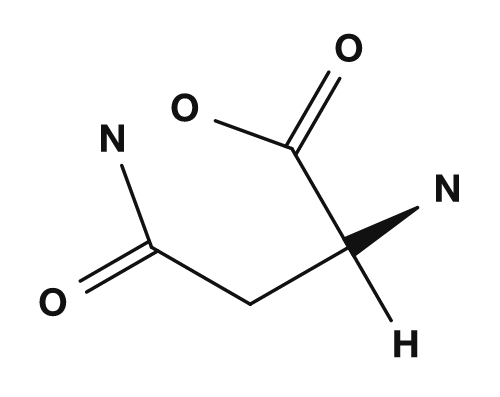
|
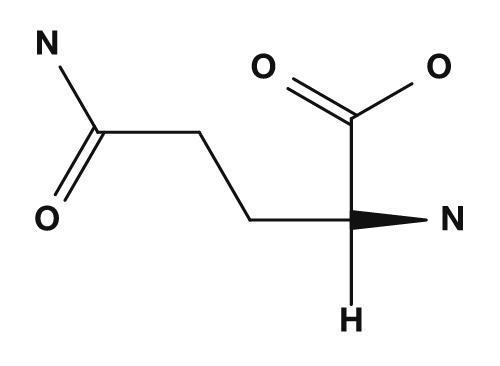
|
|
|
|
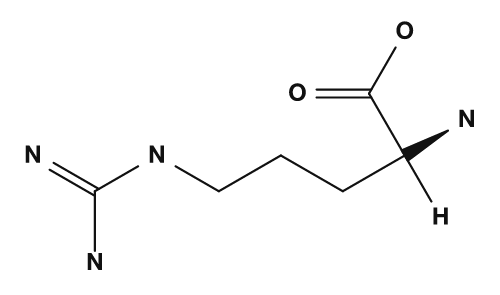
| Arginine | Histidine | Phenylalanine | Tyrosine | Tryptophan |
|---|---|---|---|---|
|
|
|
|
|
Nonpolar Amino Acids
Nonpolar amino acids include glycine, alanine, valine, leucine, isoleucine, proline, and methionine. They have side chains that do not mix well with water and tend to gather inside proteins. In particular, valine, leucine, and isoleucine are also known as branched-chain amino acids.
Polar Amino Acids (Uncharged)
Amino acids that are polar but tend not to be charged include serine, threonine, asparagine, and glutamine. They have side chains containing oxygen or nitrogen and may participate in hydrogen bonding. They interact with water at the surface of proteins and play important roles in the active sites of enzymes.
Acidic and Basic Amino Acids
Acidic Amino Acids
The acidic amino acids are aspartic acid and glutamic acid. They have carboxyl groups in their side chains and tend to carry a negative charge at physiological pH.
Basic Amino Acids
The basic amino acids are lysine, arginine, and histidine. They have nitrogen-containing side chains and exhibit basicity. In particular, lysine and arginine tend to carry a positive charge, while histidine is treated as a side chain that can be protonated depending on conditions. They are also important when considering the isoelectric point of proteins and interactions with DNA.
Aromatic Amino Acids
The aromatic amino acids are phenylalanine, tyrosine, and tryptophan. Because they have aromatic rings, they form a group that is easy to identify in structural formulas. Tyrosine has a phenolic -OH, and tryptophan has an indole ring. Aromatic amino acids are also related to hydrophobic interactions in proteins and to ultraviolet absorption.
Sulfur-Containing Amino Acids
The sulfur-containing amino acids are cysteine and methionine. Cysteine has a thiol group (-SH), and when two cysteines are oxidized they can form a disulfide bond. This bond is important for stabilizing the three-dimensional structure of proteins. Methionine has a thioether structure and is also known as a representative amino acid associated with the start codon AUG, which corresponds to the initiation of translation.
Proline: A Special Structure
Proline is somewhat special. In most amino acids, the amino group is a primary amine, but in proline the side chain returns to the amino group to form a ring, giving it a structure closer to a secondary amine. NCBI Bookshelf also explains that proline has a secondary amino group, unlike other amino acids. Because of this, proline is involved in bends in the protein main chain and in structural restrictions.
How to Read Structural Formulas
When studying the 20 amino acids through structural formulas, it is helpful to first identify the common skeleton and then compare only the R groups. Checking the following points about the R group allows you to predict the properties of an amino acid.
- Mostly hydrocarbons → hydrophobic
- Contains oxygen or nitrogen → polar
- Has an extra carboxyl group → acidic
- Has an extra amino group → basic
- Has an aromatic ring → aromatic
Summary
The 20 amino acids share the same alpha-amino acid skeleton, but their properties vary depending on differences in the side chain. Rather than memorizing only the names, organizing them under the categories of nonpolar, polar, acidic, basic, aromatic, sulfur-containing, and special structure makes it easier to read their characteristics from their structural formulas.