Most amino acids have stereoisomers. This is because the α-carbon acts as a chiral center. A chiral center is a carbon atom bonded to four different substituents. In most protein-forming α-amino acids, the α-carbon is bonded to an amino group, a carboxyl group, a hydrogen atom, and a side chain R group, making it a chiral center. NCBI Bookshelf explains that protein-forming amino acids are α-amino acids, and all except glycine have a chiral center at the α-carbon.
D and L Notation
A common notation in amino acid stereochemistry is D-form and L-form. The D/L notation represents relative configuration based on glyceraldehyde as a reference. The standard amino acids that make up proteins are fundamentally L-amino acids. NCBI Bookshelf also states that all 20 amino acids in proteins are the L-isomer.
Difference Between D/L Notation and R/S Notation
D/L notation and R/S notation are not the same thing.
| Notation System | Basis | Protein-forming Amino Acids | Exceptions |
|---|---|---|---|
| D/L notation | Relative configuration based on glyceraldehyde | L-form | Glycine (no chiral center) |
| R/S notation | Absolute configuration by CIP priority rules | Mostly S configuration | Cysteine (R configuration) |
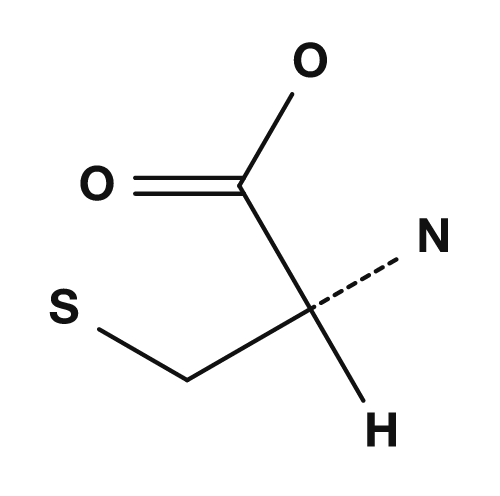
D/L is a notation based on historical relative configuration, while R/S is an absolute configuration based on CIP priority rules. Most L-amino acids have the S configuration, but cysteine is an exception — its side chain contains sulfur, which changes the priority order, giving it the R configuration. NCBI Bookshelf also states that standard amino acids except glycine and cysteine have the S configuration in R/S notation, while cysteine has the R configuration due to its sulfur-containing R group.
Identifying Chiral Centers from Structural Formulas
When looking for a chiral center in a structural formula, focus on the α-carbon. If the four substituents bonded to that carbon are all different, it is a chiral center.
| Amino Acid | Substituents at α-carbon | Chiral Center |
|---|---|---|
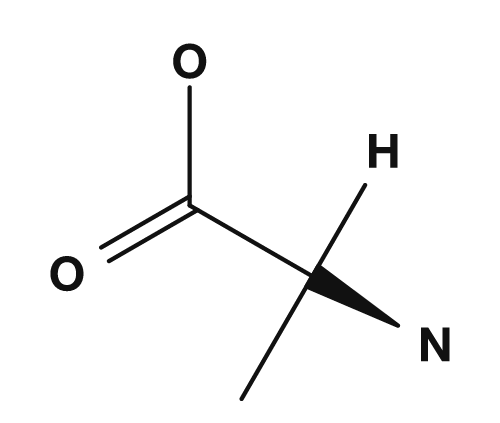
| Alanine | NH2, COOH, H, CH3 |
Yes (all four are different) |
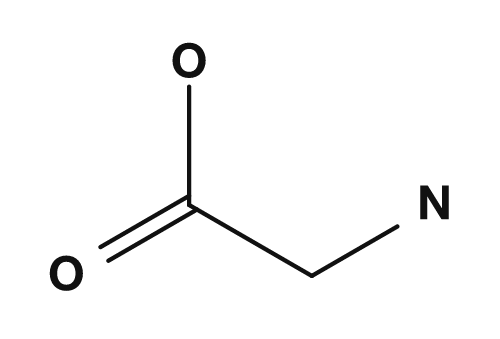
| Glycine | NH2, COOH, H, H |
No (two hydrogens) |
| Alanine | Glycine | Cysteine |
|---|---|---|
|
|
|
In alanine, the α-carbon is bonded to NH2, COOH, H, and CH3. Since all four are different, alanine is chiral. In glycine, the R group is hydrogen, so the α-carbon is bonded to NH2, COOH, H, and H — there are only three distinct substituents, so it has no chiral center. PubChem also states that glycine, with a hydrogen atom as its side chain, is the only achiral protein-forming amino acid.
D and L Forms and Optical Rotation
D-form and L-form are related as enantiomers (mirror images). When two molecules with the same molecular formula and bonding relationships cannot be superimposed on their mirror images, they are called enantiomers. In amino acids, this difference in spatial configuration greatly affects interactions with biological molecules. Since enzymes and receptors in living organisms have three-dimensional structures, they can distinguish between L and D forms.
An important point when learning D/L notation for amino acids is not to think simply that "L means levorotatory" or "D means dextrorotatory." D/L notation does not directly indicate the direction of optical rotation. Optical rotation is a property measured experimentally and expressed as + or -. D/L is a symbol representing the structural relative configuration.
Key Points for Learning from Structural Formulas
When learning from structural formulas, the first step is to check whether the α-carbon has four different substituents.
- Check whether the α-carbon has four different substituents
- Glycine is exceptionally without a chiral center
- Amino acids in proteins are fundamentally L-form
- D/L notation and R/S notation are different systems
- Cysteine is an exception: L-form but R configuration
Summary
Most amino acids are chiral because the α-carbon has four different substituents. The standard amino acids in proteins are fundamentally L-form, and only glycine lacks a chiral center because it has two identical hydrogen atoms at the α-carbon. D/L notation and R/S notation are different systems, and cysteine in particular must be understood as an exception.