The most important part to understand when learning about amino acids is the side chain. The side chain is also called the R group, and it is the part that differs between amino acids. While amino acids share a common backbone, changing the R group changes properties such as water solubility, charge, and the tendency to be buried inside a protein. NCBI Bookshelf explains that amino acids have an α-carboxyl group, an α-amino group, and an R group, and that differences in the R group determine the unique properties of each amino acid.
The basic structure of an amino acid can be represented as follows.
H2N-CH(R)-COOH
Basics of the Side Chain: Examples of R Groups
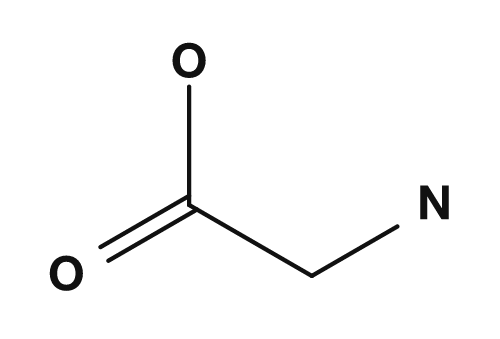
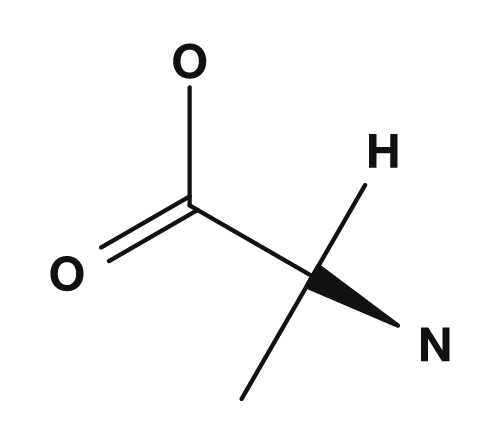
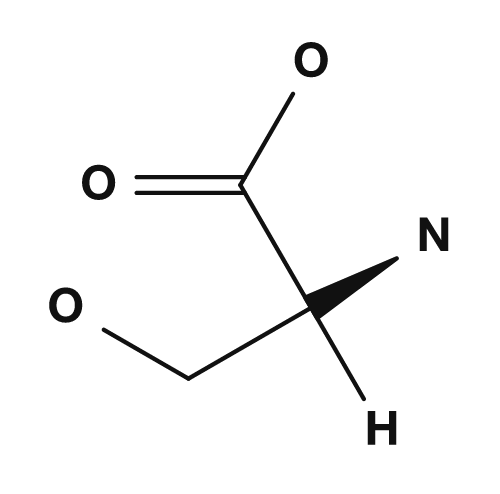
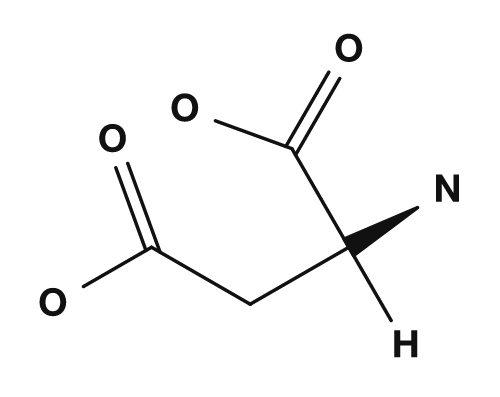
The R part is the side chain. For example, in glycine the R group is hydrogen, in alanine it is a methyl group, in serine it is a hydroxymethyl group, and in aspartic acid it is a carboxymethyl group. These small differences have a major impact on the properties of the amino acid.
| Amino acid | R group | Features |
|---|---|---|
| Glycine | Hydrogen | Smallest amino acid, achiral |
| Alanine | Methyl group | Simple nonpolar |
| Serine | Hydroxymethyl group | Polar, participates in hydrogen bonding |
| Aspartic acid | Carboxymethyl group | Acidic, tends to carry a negative charge |
| Glycine | Alanine | Serine | Aspartic acid |
|---|---|---|---|
|
|
|
|
Affinity for Water: Hydrophobic and Hydrophilic
The most fundamental way to classify side chains is by their affinity for water.
- Side chains that contain many hydrocarbons tend to be hydrophobic and tend to cluster inside the protein.
- Side chains containing oxygen or nitrogen tend to participate in hydrogen bonding and are therefore hydrophilic.
The shape of a protein is greatly influenced by these interactions among side chains. NCBI Bookshelf's Molecular Biology of the Cell explains that amino acids with hydrophobic side chains tend to gather inside the protein, and that noncovalent interactions stabilize the folded structure.
Acidic and Basic Side Chains
The next important point is acidic and basic side chains.
Aspartic acid and glutamic acid have carboxyl groups in their side chains and tend to carry a negative charge at physiological pH. Lysine, arginine, and histidine have basic nitrogen in their side chains. In particular, lysine and arginine tend to carry a positive charge, and histidine is important as a side chain that is easily protonated depending on conditions. A paper published on PMC also explains that, around neutral pH, aspartic acid and glutamic acid carry negative charges, and lysine, arginine, and histidine are treated as amino acids involved in the charge state.
| Charge | Representative amino acids | Functional groups in the side chain |
|---|---|---|
| Negative (acidic) | Aspartic acid, glutamic acid | Carboxyl group |
| Positive (basic) | Lysine, arginine, histidine | Amino group, guanidino group, imidazole |
| Lysine | Phenylalanine | Cysteine |
|---|---|---|
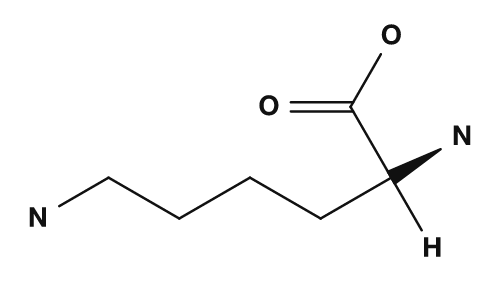
|
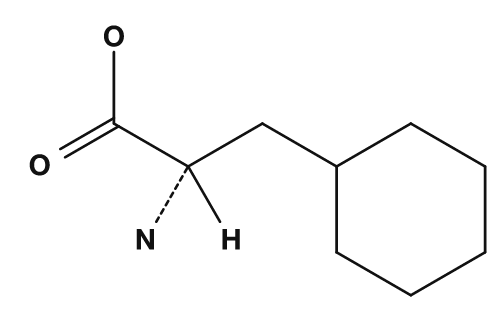
|
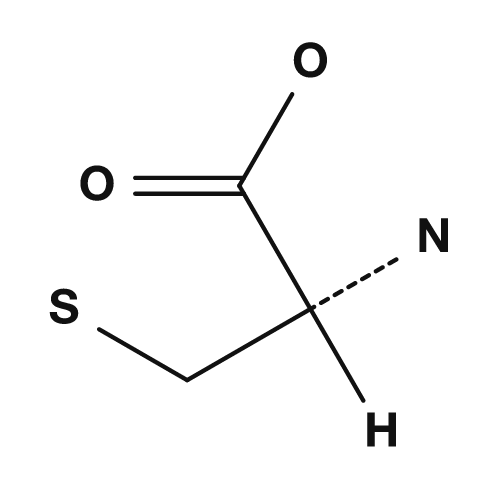
|
Aromatic Side Chains
Amino acids with aromatic side chains include phenylalanine, tyrosine, and tryptophan. These have cyclic π electron systems and are relatively easy to spot in structural formulas. Tyrosine has a phenolic hydroxyl group, so it shows different properties from a simple hydrophobic aromatic. Tryptophan has a large indole ring and is also important from the standpoint of protein structure and light absorption.
Sulfur-Containing Side Chains
Sulfur-containing side chains also deserve attention.
Cysteine has a thiol group (-SH), and when two cysteines are oxidized, they can form a disulfide bond. This is an important bond that stabilizes the three-dimensional structure of proteins. Methionine has a thioether structure and is also well known as the amino acid corresponding to the start codon AUG for translation initiation.
| Side chain type | Representative amino acids | Key points |
|---|---|---|
| Hydrophobic | Valine, leucine, isoleucine | Tend to cluster inside proteins |
| Aromatic | Phenylalanine, tyrosine, tryptophan | π electron system, involved in UV absorption |
| Sulfur-containing | Cysteine, methionine | Disulfide bonds, relationship to start codon AUG |
How to Read Structural Formulas
When learning about side chains, it is easier to understand if you think from the structural formula rather than memorizing amino acid names and properties separately.
- Has oxygen or nitrogen → tends to be polar
- Has a carboxyl group → tends to be acidic
- Has an amino or guanidino group → tends to be basic
- Has many hydrocarbons → tends to be hydrophobic
Summary
The side chain of an amino acid is the key part that determines its properties. Differences in the R group give rise to features such as hydrophobicity, hydrophilicity, acidity, basicity, aromaticity, and sulfur content. When looking at structural formulas, paying attention not only to the common backbone but also to what atoms and functional groups are in the side chain makes it much easier to understand the properties of amino acids.