Amino acids change their charge state depending on pH. Under acidic conditions they tend to carry a positive charge, and under basic conditions a negative charge. The isoelectric point is key to understanding these charge changes. The isoelectric point can be understood as the pH at which the net charge of the molecule becomes zero, with positive and negative charges in balance. The IUPAC Gold Book describes the isoelectric state as the condition in which a polyampholyte, especially a protein, shows no electrophoresis.
pH and Charge Changes
The basic structure of amino acids includes an amino group and a carboxyl group.
- Under acidic conditions, the amino group tends to accept a proton and become
-NH3+, while the carboxyl group tends to take the form-COOH. - Under basic conditions, the carboxyl group loses a proton and becomes
-COO-, and the amino group approaches the-NH2state. - At intermediate pH, the form with both
-NH3+and-COO-(zwitterion) predominates.
| pH Condition | Amino Group State | Carboxyl Group State | Net Charge |
|---|---|---|---|
| Acidic (low pH) | -NH3+ (positive) |
-COOH (neutral) |
Positive |
| Near isoelectric point | -NH3+ (positive) |
-COO- (negative) |
0 (zwitterion) |
| Basic (high pH) | -NH2 (neutral) |
-COO- (negative) |
Negative |
What Is the Isoelectric Point?
Taking glycine, the simplest amino acid, as an example: on the acidic side it tends to carry an overall positive charge, and on the basic side a negative charge. Somewhere in between, there is a pH at which the net charge becomes zero. This is glycine's isoelectric point.
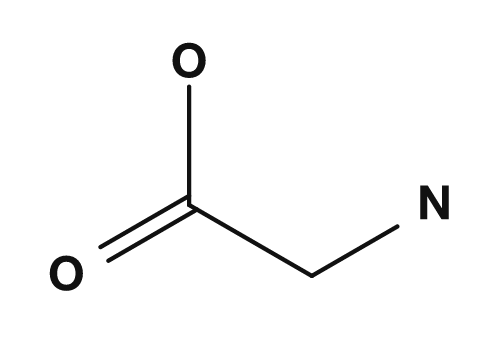
When understanding the isoelectric point, it is more accurate to think of it as a state where positive and negative charges are balanced rather than a state with no charges at all. Amino acids tend to exist as zwitterions near neutral pH, carrying both positive and negative charges within the molecule. PubChem explains that the zwitterion of L-cysteinylglycine arises from the transfer of a proton from a hydroxy group to an amino group, and is the major microspecies at pH 7.3.
How Side Chains Affect the Isoelectric Point
The isoelectric point varies depending on the type of side chain.
- Aspartic acid and glutamic acid, which have carboxyl groups in their side chains, tend to carry a negative charge, so their isoelectric points are lower.
- Amino acids with basic side chains, such as lysine and arginine, tend to carry a positive charge, so their isoelectric points are higher.
A PMC article explains that the pI of a protein is defined as the pH at which the net charge is zero, and depends strongly on the amino acid composition and the pKa values of each side chain.
| Side Chain Nature | Representative Amino Acids | Effect on Isoelectric Point |
|---|---|---|
| Side chain with carboxyl group (acidic) | Aspartic acid, Glutamic acid | Lowers the isoelectric point |
| Side chain with basic nitrogen (basic) | Lysine, Arginine, Histidine | Raises the isoelectric point |
| Aspartic acid | Glutamic acid | Lysine |
|---|---|---|
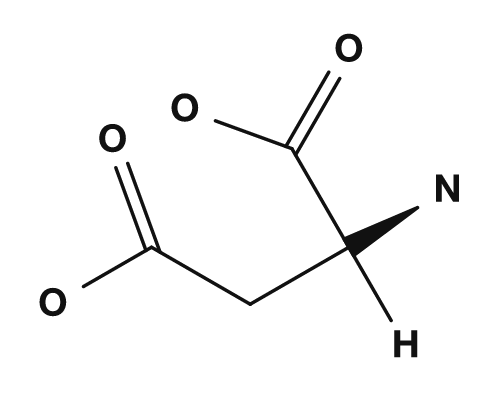
|
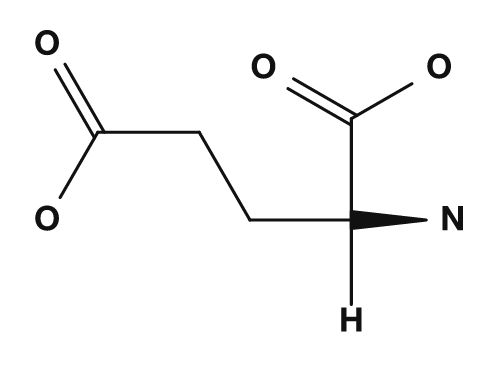
|
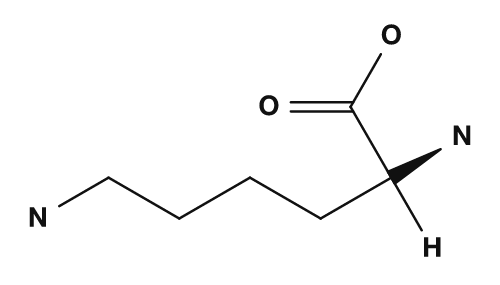
|
| Arginine | Histidine |
|---|---|
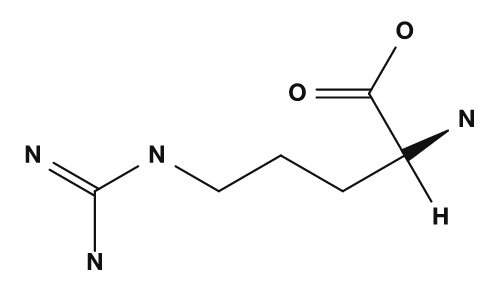
|
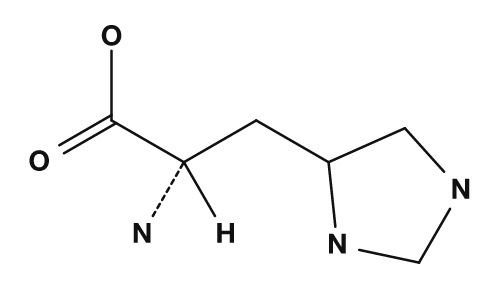
|
Thinking About Charge from Structural Formulas
When thinking about the isoelectric point from structural formulas, the first step is to look for parts that can carry a charge.
- Identify the amino group and carboxyl group
- Check whether the side chain has acidic or basic functional groups
- Aspartic acid and glutamic acid → carboxyl group in side chain
- Lysine → amino group in side chain
- Arginine → guanidino group
- Histidine → imidazole ring
Applications of the Isoelectric Point
The isoelectric point is also important in the separation and purification of proteins. Because the net charge of a molecule changes with pH, it is used in electrophoresis and ion-exchange chromatography.
- When pH is below the isoelectric point → the molecule tends to be positively charged
- When pH is above the isoelectric point → the molecule tends to be negatively charged
A PMC article also states that proteins carry a positive charge at pH below pI and a negative charge at pH above pI.
Summary
The isoelectric point is the pH at which the net charge of an amino acid or protein becomes zero. In amino acids, the ionization states of the amino group, carboxyl group, and side chain change with pH, so the charge also changes. When reading structural formulas, looking for functional groups that can carry a charge is the first step to understanding the isoelectric point.