Amino acids are the basic units that make up proteins. In living organisms, when proteins are broken down they become amino acids, and those amino acids are then used again to synthesize proteins, repair body tissues, and support metabolism. MedlinePlus explains that amino acids and proteins are fundamental building blocks of life, and that the body uses amino acids to make proteins.
Looking at their chemical structure, amino acids are, as their name suggests, compounds that have an amino group and a carboxyl group. A typical amino acid has a central carbon atom bonded to an amino group, a carboxyl group, a hydrogen atom, and a side chain. This side chain is also called the R group and differs from one amino acid to another. NCBI Bookshelf explains that the amino acids that make up proteins have an α-carboxyl group, an α-amino group, and an R group, and that differences in the R group determine the properties of each amino acid.
The Basic Chemical Structure
The basic structure of an amino acid can be represented by the following structural formula.
H2N-CH(R)-COOH
The parts of this formula correspond to the following.
| Symbol | Name |
|---|---|
CH |
alpha carbon |
NH2 |
amino group |
COOH |
carboxyl group |
R |
side chain (different for each amino acid) |
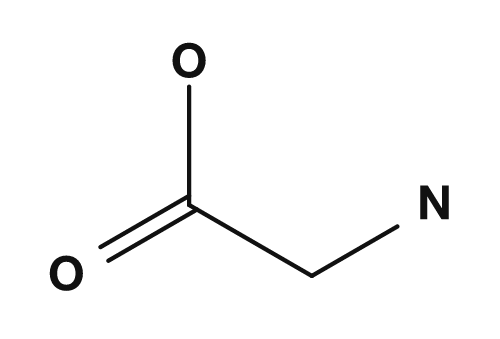
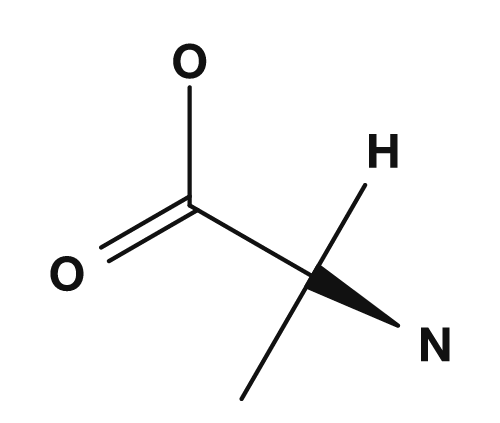
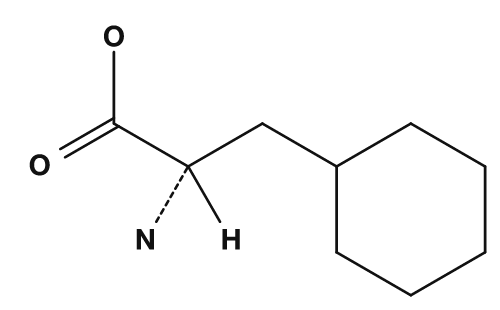
The type of amino acid is determined by the R group. If R is hydrogen, the amino acid is glycine; if it is a methyl group, alanine; if it is a benzyl group, phenylalanine. In other words, even though the common part is the same, changing the R group produces a different amino acid.
| Amino acid | R group |
|---|---|
| Glycine | Hydrogen |
| Alanine | Methyl group |
| Phenylalanine | Benzyl group |
| Glycine | Alanine | Phenylalanine |
|---|---|---|
|
|
|
The Side Chain (R Group) Determines Properties
When understanding amino acids through their structural formulas, the important point is not simply to memorize them as "the building blocks of proteins." The amino group tends to be basic, the carboxyl group tends to be acidic, and the side chain determines properties such as hydrophobicity, hydrophilicity, acidity, basicity, and aromaticity. Therefore, by looking at the structure of an amino acid, you can get clues as to whether it tends to dissolve in water, whether it tends to be buried inside a protein, or whether it tends to carry a charge.
For example, glycine has only hydrogen as its side chain, making it the smallest amino acid. PubChem also describes glycine as the simplest proteinogenic amino acid, with a hydrogen atom as its side chain, and as the only achiral proteinogenic amino acid. On the other hand, amino acids with basic side chains such as lysine and arginine tend to carry a positive charge under physiological conditions and are involved in interactions on the surface of proteins and with other molecules.
Four Elements for Understanding the Structure
When learning about amino acids, rather than suddenly memorizing all 20 names, it is more important to first understand the common skeleton. Once you can identify the following four elements in a structural formula, you can begin to classify each amino acid based on its structure.
- Amino group (
-NH2) - Carboxyl group (
-COOH) - Alpha carbon (the central carbon)
- R group (side chain)
Summary
Amino acids are organic compounds that have an amino group and a carboxyl group. In the amino acids that make up proteins, an amino group, a carboxyl group, a hydrogen atom, and an R group are bonded to the alpha carbon. Differences between amino acids are determined mainly by the R group, and these differences also influence the properties and functions of proteins.