There are many kinds of amino acids, but most of the major amino acids that make up proteins are alpha amino acids. An alpha amino acid is an amino acid in which the amino group and the carboxyl group are bonded to the same carbon, that is, to the alpha carbon. NCBI Bookshelf explains that the alpha amino acids that make up proteins have their amino group and carboxylic acid functional group separated by a single carbon atom, and they typically have a chiral carbon.
The Structure of an Alpha Amino Acid
First, consider the carbon of the carboxylic acid as a reference. The carbon next to the carboxyl group -COOH is the alpha carbon. An amino acid in which the amino group -NH2 is bonded to that alpha carbon is an alpha amino acid. The structural formula can be written as follows.
H2N-CH(R)-COOH
Here, the carbon of CH(R) is the alpha carbon. Four groups are bonded to this carbon: the amino group, the carboxyl group, a hydrogen atom, and the R group. Differences in the R group give rise to glycine, alanine, valine, phenylalanine, and so on.
| Glycine | Alanine |
|---|---|
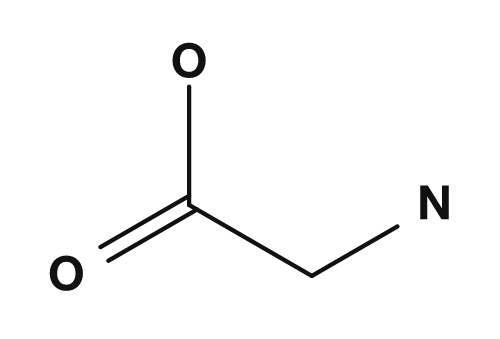
|
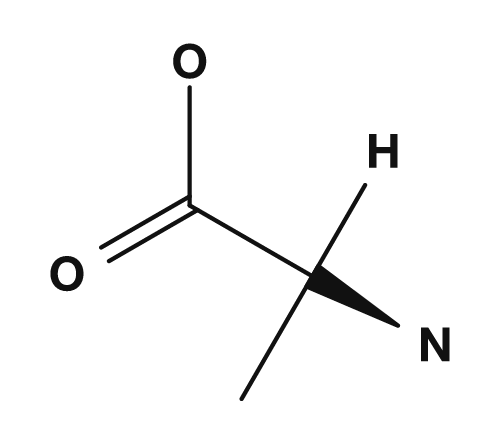
|
Why Is It Called "Alpha"?
In organic chemistry, the carbon next to a particular functional group is sometimes called the alpha carbon, the next one the beta carbon, and the one after that the gamma carbon. In the case of amino acids, the amino group is bonded to the alpha carbon, which is right next to the carboxyl group, so they are called alpha amino acids.
| Position of the amino group | Name |
|---|---|
| Alpha carbon (next to the carboxyl group) | alpha amino acid |
| Beta carbon (two carbons away) | beta amino acid |
| Gamma carbon (three carbons away) | gamma amino acid |
Protein Structure and Alpha Amino Acids
The fact that the amino acids making up proteins are alpha amino acids is important when considering protein structure. Because the amino side and the carboxyl side around the alpha carbon are connected by peptide bonds, a regular repeating structure is created in the main chain of the protein. NCBI Bookshelf explains that proteins are polypeptides in which amino acids are connected by peptide bonds, and that the amino acid sequence determines the primary structure of the protein.
How to Recognize Them in a Structural Formula
When looking at an alpha amino acid in a structural formula, the following three points make it easier to identify.
- Find the carboxyl group
-COOH. - Identify the carbon next to it (the alpha carbon).
- Check whether the amino group is bonded to that alpha carbon.
These three steps allow you to determine whether a compound is an alpha amino acid.
Relationship to Chirality
In most alpha amino acids, the alpha carbon is a chiral center. Since four different substituents are bonded to the alpha carbon, stereoisomers exist. However, only glycine, whose R group is hydrogen, has two hydrogen atoms attached to the alpha carbon and therefore does not have a chiral center. This point is very important when learning about the stereochemistry of amino acids. PubChem also describes glycine as the only achiral proteinogenic amino acid.
Summary
An alpha amino acid is an amino acid in which the amino group is bonded to the alpha carbon, located next to the carboxyl group. The standard amino acids that make up proteins are mainly alpha amino acids, and this common skeleton is the basis for peptide bonds and the primary structure of proteins. When looking at a structural formula, it is important to check the positional relationship of the carboxyl group, the alpha carbon, and the amino group.