Amino acids do not exist only on their own; they link together to form peptides and proteins. The bond that connects amino acids in this case is the peptide bond. According to NCBI Bookshelf, proteins are chains of amino acids linked by peptide bonds, and the amino acid sequence determines the primary structure.
| Glycine | Alanine |
|---|---|
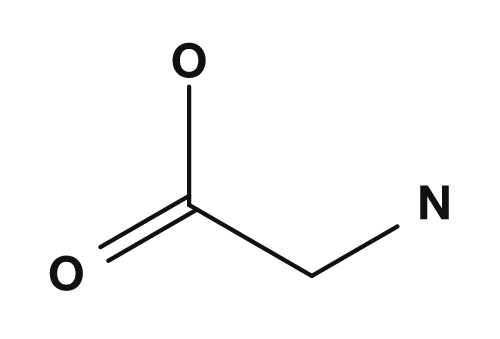
|
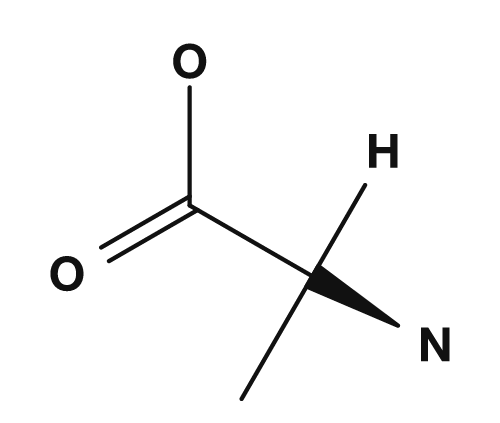
|
Formation of the Peptide Bond
A peptide bond is an amide bond formed when the carboxyl group of one amino acid reacts with the amino group of another amino acid. The reaction releases one molecule of water, so it is described as a condensation reaction. Expressed as a structural formula, it produces the following partial structure.
-CO-NH-
This -CO-NH- is the peptide bond. Since it is the structure formed when a carboxylic acid and an amine combine to give an amide, the peptide bond can be regarded as a type of amide bond. In the Peptide entry of NCBI Bookshelf, peptides are described as short chains of 2–50 amino acids covalently linked by condensation reactions.
Types of Peptide Chains
The naming changes depending on the number of amino acids that are linked. In Molecular Biology of the Cell, proteins are described as long chains of 20 different amino acids linked by covalent peptide bonds.
| Name | Number of amino acids |
|---|---|
| Dipeptide | 2 |
| Tripeptide | 3 |
| Oligopeptide | A few to about a dozen |
| Polypeptide | More than that |
| Protein | Long polypeptide chains that have folded |
A Key Feature of Peptide Bonds: Partial Double-Bond Character
An important feature of the peptide bond is that, unlike an ordinary single bond, it has partial double-bond character. This is because the lone pair of electrons on the amide nitrogen undergoes resonance with the carbonyl group. As a result, the area around the peptide bond has planarity, and rotation is restricted. This property is very important when considering the three-dimensional structure of proteins.
Main features of the peptide bond:
- A type of amide bond (
-CO-NH-) - Formed by a condensation reaction (loss of a water molecule)
- Has partial double-bond character
- The region around the bond has planarity, with restricted rotation
N-terminus and C-terminus
A peptide chain has directionality.
| Terminus | Remaining functional group | Reading convention |
|---|---|---|
| N-terminus | Amino group (-NH2) |
Start of the amino acid sequence |
| C-terminus | Carboxyl group (-COOH) |
End of the amino acid sequence |
The amino acid sequence of a protein is usually written from the N-terminus to the C-terminus. Understanding this directionality makes it easier to read the structural formulas of peptides and proteins.
Identifying Peptide Bonds in Structural Formulas
To identify a peptide bond in a structural formula, look for the -CO-NH- portion. Since the carbonyl carbon is bonded to a nitrogen, it has the same structure as an amide. However, when the α-carbons derived from amino acids are connected on both sides, it is treated as a peptide bond.
Biological Importance
The peptide bond is extremely important in living organisms. Many biological molecules, such as enzymes, antibodies, hormones, and structural proteins, are formed by linking amino acids through peptide bonds. MedlinePlus also explains that amino acids form proteins and are used for the growth and repair of body tissues.
Summary
A peptide bond is the -CO-NH- bond formed by condensation between the carboxyl group of one amino acid and the amino group of another. The peptide bond is a type of amide bond and has partial double-bond character, which also affects the three-dimensional structure of proteins. By linking amino acids through peptide bonds, peptides and proteins are formed.